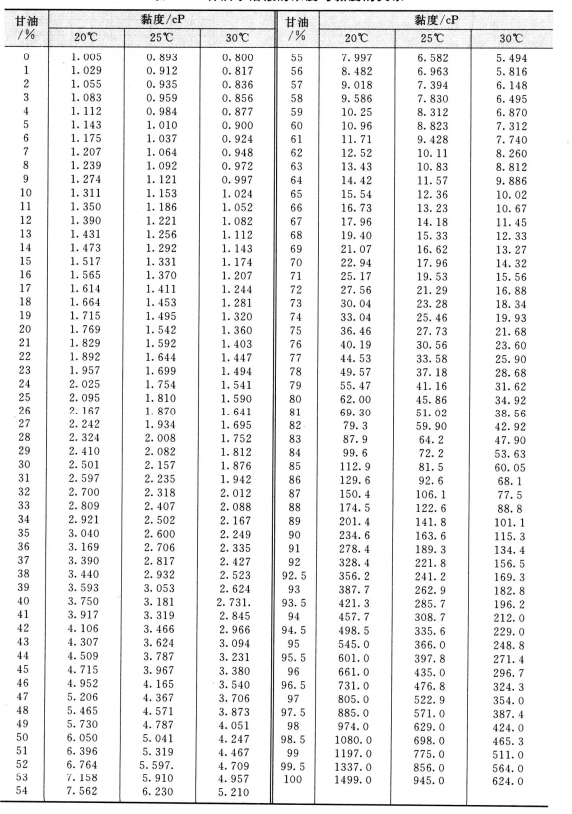
The viscosity of water decreases smoothly from the freezing point while the density remains essentially constant except for the small maximum in density near 4☌. In cgs units the unit is called the centistoke. each sample was taken at a temperature of 25C which is the standard ambient. The kinematic viscosity is the dynamic viscosity divided by the density of the fluid. composition of xanthan gum-starch-water-glycerol-contrast agent mixture. The unit centiPoise = 0.001 Pa s and is is sometimes written mPa s. Bui and Nguyen4 adjusted an empirical equation to viscosities of glucoseaqueous solutions as a function of temperature. See Figure 1-3 for vapor pressure curve Viscosity at 25 C (77 F): my ex best. Viscosities were determined at 25☌ in the molality range of 0.1 to 1.8 moles/kg(3.4 to 61.6 w/w) for sucrose, and in the range of 1.2 to 7.5 moles/kg (21.6 to 135.1 w/w)for glucose. These viscosities are at 20☌ except for the blood and blood plasma which are at body temperature, 37☌, and for steam which is at 100☌. Kinematic viscosity at 40 ☌ and 100 ☌ - low-shear viscosity - determined according to ASTM D445. The Poise is used in the table because of its more common usage. More commonly used is the dyne sec/cm 2 which is called Poise. 0.5 Mol of bromoalkane (CiH2i+1Br) was then added drop wise. Viscosities were determined at 25☌ in the molality range of 0.1 to 1.8 moles/kg (3.4 to 61.6 w/w) for sucrose, and in the range of 1.2 to 7.5 moles/kg. Viscosity has the SI units Pascal seconds (Pa s) which is called the Poiseuille. A solvent or composition that includes a glycerin short-chain aliphatic ether may. A similar work was carried out with sucrose solutions and kinematic viscosity data were reported from 20 to 50☌ and molalities in the range of 0.5 to 4.5 moles/kg (14.61 to. Viscosity of Liquids and Gases Viscosity of Liquids and Gases Liquids
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
